Immersive Training for Life Sciences & Pharma
Train lab technicians, manufacturing operators, clinical teams, and field representatives with interactive 3D simulations — built for the precision, compliance, and global scale that life sciences demands.
Life Sciences Training Applications
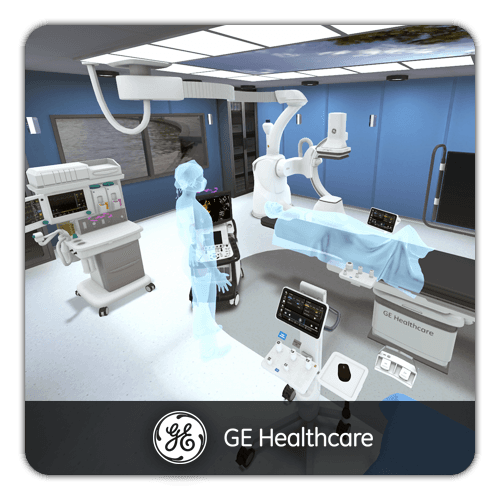
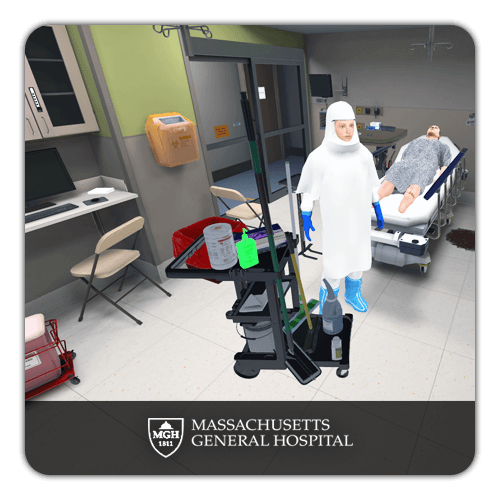
GMP and Cleanroom Compliance Training
Good Manufacturing Practice (GMP) compliance is non-negotiable in life sciences. Deploy standardized GMP training across all manufacturing sites with interactive 3D cleanroom environments. Learners practice gowning procedures, contamination prevention protocols, and cleanroom behavior in realistic simulations. Built-in assessments verify individual proficiency and generate documentation for regulatory audits.
Laboratory Procedures and Equipment Training
Train lab technicians on analytical instruments, bioprocessing equipment, and standard operating procedures using interactive 3D replicas. Learners practice sample preparation, instrument calibration, and multi-step protocols hands-on — reducing errors when they transition to the real lab. Particularly valuable for expensive or limited-access instruments where hands-on practice time is scarce.
Drug Manufacturing and Bioprocessing
Pharmaceutical manufacturing involves complex, multi-step processes where errors can destroy entire batches worth millions. VR training lets operators practice batch manufacturing, bioreactor operation, fill-finish procedures, and quality control processes in virtual environments that replicate production conditions without consuming real materials.
Clinical Training and MSL Education
Train Medical Science Liaisons (MSLs) and clinical teams on mechanisms of action, clinical trial data, and therapeutic area knowledge using interactive 3D molecular and anatomical visualizations. Transform complex scientific data into experiential learning that builds deeper understanding than slides or publications.
Field Representative and Sales Training
Equip pharmaceutical field representatives with interactive 3D product demonstrations they can present to healthcare providers on any device. Reps practice product presentations, objection handling, and clinical data discussions in simulated scenarios before engaging real customers.
Standard Operating Procedure (SOP) Training
SOPs are the backbone of life sciences operations, and keeping training current with frequent SOP revisions is a constant challenge. No-code authoring tools let your training team update SOP training modules in hours — not the weeks or months required for traditional eLearning redevelopment.
Why Life Sciences Companies Choose EducationXR
Regulatory-Ready Assessments
Life sciences training isn't optional — it's auditable. Built-in assessment builders track procedural accuracy, completion status, and individual proficiency scores. Export assessment data in formats suitable for FDA, EMA, and other regulatory submissions.
Global Deployment, Instant Updates
Pharma companies operate manufacturing sites, labs, and commercial teams across dozens of countries. Cross-platform deployment reaches every employee on VR headsets, tablets, or desktops from a single content build. When SOPs change, update the training once and it propagates globally within minutes.
Real-Time 3D for Molecular Visualization
Interactive 3D models of molecules, proteins, anatomical structures, and medical devices deliver understanding that 2D diagrams and slide decks cannot match. Learners rotate, zoom, disassemble, and explore complex structures at their own pace.
Automatic Multiplayer for Instructor-Led Training
Remote instructor-led training sessions are increasingly common in global life sciences organizations. Automatic multiplayer means an instructor can guide learners through a procedure in a shared virtual space — with participants joining from VR headsets, tablets, or laptops across multiple countries.
No-Code Content Creation for SMEs
Regulatory affairs specialists, quality managers, and scientific trainers understand the content better than any external developer. FLOW visual scripting lets them build and update interactive training independently, keeping content current with rapidly evolving procedures and regulations.
Enterprise Security and Administration
SSO integration, user and group management, role-based content access, branded distribution, and a 99.5% uptime SLA. Built for the compliance and security requirements of regulated industries.
$1,200-$1,800
Average annual training spend per employee in pharma
$10M+
Average cost per FDA warning letter for GMP violations
80%
Knowledge retention at 1 year with VR training
40-75%
Reduction in onboarding time with VR manufacturing training
Life Sciences Training FAQ
Common questions from life sciences, pharmaceutical, and biotech training leaders.
Which life sciences and pharmaceutical companies use EducationXR?
EducationXR is used by Merck, Thermo Fisher Scientific, Edwards Lifesciences, Bracco, Terumo, and other pharmaceutical and medical device companies for GMP compliance, lab training, clinical education, and field representative enablement. Applications span drug manufacturing operator training, analytical instrument operation, medical device training, and MSL education.
Is EducationXR suitable for GMP and cleanroom training?
Yes. EducationXR delivers interactive 3D cleanroom environments where operators practice gowning procedures, contamination prevention protocols, and cleanroom behavior in realistic virtual simulations. Built-in assessments track procedural accuracy and individual proficiency, and exportable data supports FDA, EMA, and other regulatory audit documentation. The cost of a single FDA warning letter for GMP violations typically exceeds $10M — rigorous training is a direct risk-reduction lever.
How quickly can training modules be updated when SOPs change?
SOPs change frequently in life sciences, and keeping training current is a constant challenge. FLOW visual scripting and drag-and-drop authoring inside Unity Editor let your training team update procedural modules in hours — not the weeks or months required for traditional eLearning redevelopment. A typical procedural update can be authored in as little as 15 minutes, and updates propagate immediately to all deployed devices via cloud distribution.
Can we use VR training to reduce the cost of bioprocessing operator errors?
Yes. Pharmaceutical manufacturing batches can be worth millions, and operator errors in fill-finish, bioreactor operation, or batch manufacturing can destroy entire batches. VR training lets operators practice multi-step procedures in virtual environments that replicate production conditions — without consuming real materials or compromising batches. Published outcomes for VR-based procedural training include a 3x improvement in knowledge retention and 70% reduction in time-to-proficiency.
Does EducationXR support MSL and field representative training?
Yes. Medical Science Liaisons and pharmaceutical field reps use EducationXR to train on mechanisms of action, clinical trial data, and therapeutic area knowledge using interactive 3D molecular and anatomical visualizations. Reps can also present the same 3D content to healthcare providers on any device — phone, tablet, laptop, or headset — transforming product detailing from static slide decks to interactive exploration.
Can we deploy training globally to our manufacturing sites and commercial teams?
Yes. A single content build deploys to VR headsets, tablets, phones, and desktops simultaneously. When SOPs or therapeutic content changes, update the training once and it propagates globally within minutes. EducationXR supports SSO integration, role-based content access, branded content distribution, and a 99.5% uptime SLA — built for the compliance and scale requirements of global life sciences organizations.
Who builds the training content — our SMEs or external developers?
Your subject-matter experts. Regulatory affairs specialists, quality managers, and scientific trainers understand the content better than any external developer, and FLOW visual scripting lets them build and update interactive training independently — no Unity or developer experience required. This keeps training current with rapidly evolving procedures and regulations, and reduces ongoing content-development cost.
Accelerate Life Sciences Training
EducationXR powers immersive training for some of the world's leading life sciences organizations. From GMP compliance to clinical education to field sales enablement, our platform helps you train with precision, measure with confidence, and scale globally.